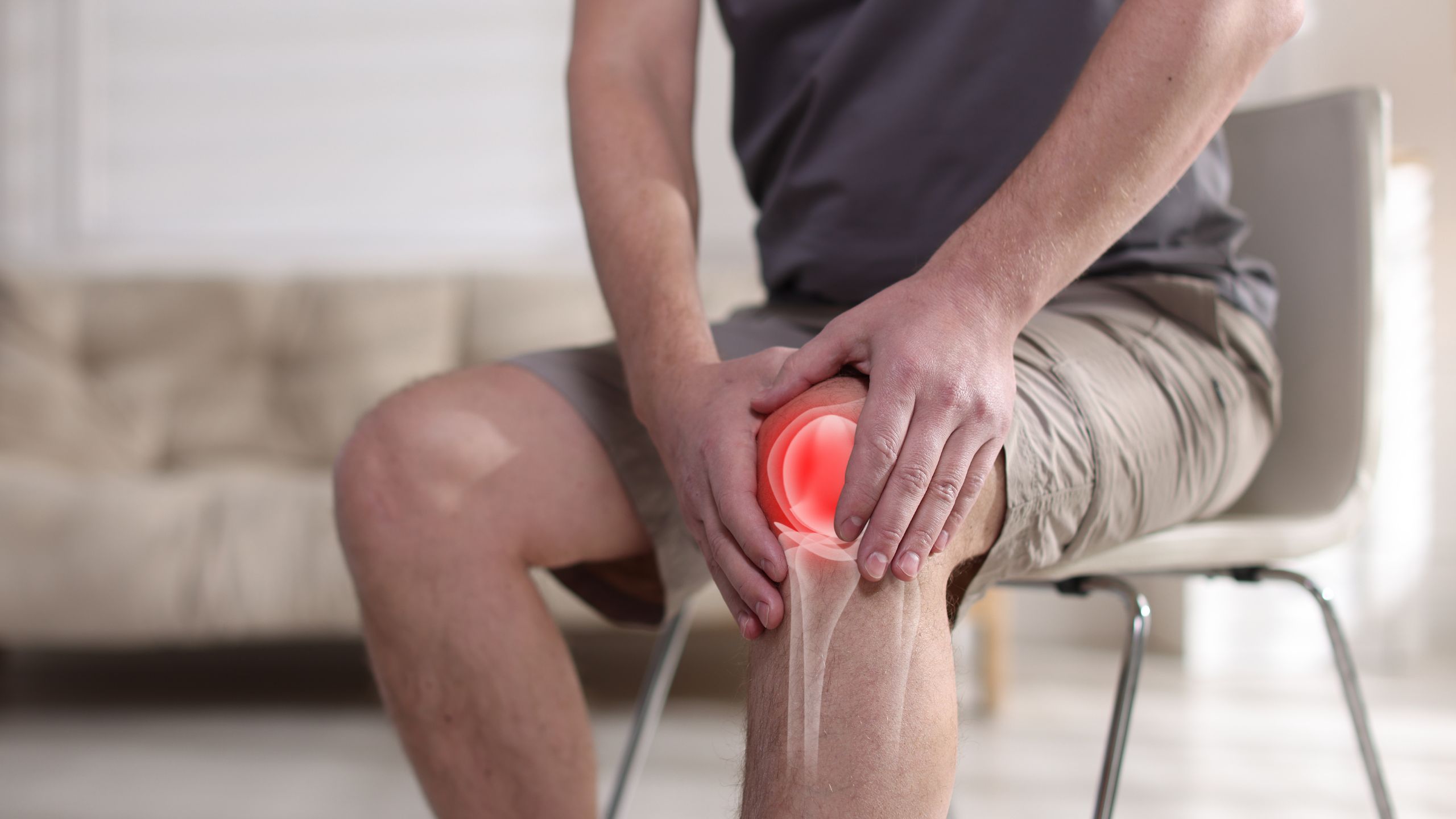
Inflammation has become a health buzzword, blamed for everything from joint pain to brain fog to weight gain. But beneath the wellness industry hype lies a fundamental biological truth: chronic low-grade inflammation is the common mechanism driving virtually every major age-related disease.
This isn’t speculation or alternative medicine theory. The role of inflammation in disease development is one of the most well-established findings in modern medicine, supported by thousands of studies across multiple fields. Chronic inflammation increases diabetes risk, accelerates cardiovascular disease, promotes cancer development, drives neurodegenerative conditions like Alzheimer’s, and literally speeds up the aging process at the cellular level.
Yet most people have no idea whether they’re inflamed, what’s causing it, or what to do about it. Standard healthcare rarely measures inflammatory status unless you already have obvious disease. By the time inflammation becomes symptomatic—joint pain, autoimmune conditions, cardiovascular events—it has been silently damaging your body for years or decades.
Understanding inflammation—what it is, what causes it, how to measure it, and most importantly how to reduce it—is essential for anyone serious about disease prevention and longevity. Inflammation isn’t just about feeling bad today. It’s about the trajectory of your health over decades.
This guide cuts through the noise to explain what inflammation actually is, why chronic inflammation is so harmful, what biomarkers reveal inflammatory status, and what evidence-based interventions actually reduce inflammatory burden and disease risk.
What Is Inflammation? Acute vs. Chronic
Inflammation is your immune system’s response to perceived threats—infections, injuries, toxins, or cellular damage. Acute inflammation is beneficial, even essential for survival. When you cut your finger, inflammation brings immune cells to fight potential infection and begin healing. When you have the flu, inflammation helps eliminate the virus. The heat, redness, swelling, and pain of acute inflammation signal that your immune system is responding appropriately.
Acute inflammation is self-limiting. Once the threat is eliminated and healing is complete, inflammatory signals resolve. Anti-inflammatory processes dominate, clearing inflammatory molecules and returning the body to homeostasis. This cycle—activation, response, resolution—is how inflammation is supposed to work.
Chronic inflammation is fundamentally different. Instead of a brief response to a specific threat followed by resolution, chronic inflammation persists at low levels indefinitely. There’s no clear infection or injury driving it, just ongoing activation of inflammatory pathways at levels too low to cause obvious symptoms but high enough to cause cumulative cellular damage over years.
This persistent low-grade inflammation goes by various names in medical literature—chronic inflammation, systemic inflammation, metaflammation (metabolic inflammation), or “inflammaging” when referring to age-related inflammation. Regardless of terminology, the pattern is the same: sustained elevation of inflammatory markers including C-reactive protein, interleukin-6, tumor necrosis factor-alpha, and others.
You can’t feel chronic inflammation the way you feel acute inflammation. There’s no obvious redness or swelling or heat. Most people with significantly elevated inflammatory markers feel essentially normal until the cumulative damage manifests as diagnosable disease—a heart attack, diabetes diagnosis, cancer, or cognitive decline.
This is what makes chronic inflammation so insidious. It operates silently for decades, incrementally damaging vascular endothelium, promoting insulin resistance, interfering with cellular signaling, and creating conditions favoring disease development. By the time symptoms appear, substantial damage has occurred.
The goal of anti-inflammatory lifestyle interventions isn’t eliminating inflammation—that would impair immune function and healing. It’s preventing chronic inflammation from establishing and persisting over time. It’s maintaining the inflammatory system’s ability to activate when needed and, critically, to resolve completely rather than smoldering indefinitely.
How Chronic Inflammation Drives Disease
Chronic inflammation doesn’t just correlate with disease—it directly causes and accelerates disease development through well-characterized mechanisms.
Cardiovascular disease is fundamentally an inflammatory condition. Atherosclerosis—the buildup of plaque in arteries leading to heart attacks and strokes—begins with inflammation in the arterial wall. Inflammatory molecules damage vascular endothelium, making it permeable to LDL cholesterol. Immune cells infiltrate the vessel wall, engulf oxidized LDL, and form foam cells that become atherosclerotic plaque. Chronic inflammation accelerates every stage of this process.
C-reactive protein (CRP)—the primary clinical marker of inflammation—predicts cardiovascular events as strongly as LDL cholesterol. People in the highest quartile of CRP have double the heart attack risk of those in the lowest quartile, independent of cholesterol levels. The CANTOS trial proved causation: reducing inflammation with an anti-inflammatory drug (without affecting cholesterol) reduced cardiovascular events by 15%, confirming that inflammation itself drives cardiovascular disease.
Type 2 diabetes is increasingly understood as an inflammatory disease. Chronic inflammation interferes with insulin signaling, creating insulin resistance. Fat tissue, particularly visceral fat, produces inflammatory cytokines—TNF-alpha, IL-6, resistin—that block insulin receptors and impair glucose uptake. The inflammatory state also affects pancreatic beta cells, reducing insulin secretion capacity.
Elevated inflammatory markers predict diabetes development years before diagnosis. Someone with elevated CRP today has 2-3 times higher diabetes risk over the next decade compared to someone with low CRP, even with identical weight, diet, and exercise patterns. Inflammation precedes and causes metabolic dysfunction.
Cancer development is promoted by chronic inflammation through multiple pathways. Inflammation causes DNA damage through reactive oxygen species and other mutagens. It promotes cellular proliferation while impairing apoptosis (programmed cell death), allowing damaged cells to survive and multiply. It creates a tissue environment supporting tumor growth and metastasis.
About 20% of cancers are directly linked to chronic infections causing inflammation—H. pylori causing stomach cancer, hepatitis B and C causing liver cancer, HPV causing cervical cancer. But even cancers without infectious causes develop more readily in inflamed tissue. Inflammatory bowel disease dramatically increases colon cancer risk. Chronic inflammation in other tissues similarly increases cancer susceptibility.
Neurodegenerative diseases including Alzheimer’s disease are characterized by chronic brain inflammation. Microglial cells—the brain’s immune cells—become chronically activated, producing inflammatory cytokines that damage neurons and promote tau protein tangles and amyloid plaque formation. Elevated systemic inflammation measured in blood correlates with faster cognitive decline and increased dementia risk.
Accelerated aging at the cellular level is driven by inflammation. Inflammatory stress shortens telomeres—the protective caps on chromosomes that prevent DNA damage during cell division. Short telomeres indicate advanced biological age and predict mortality. Chronic inflammation also promotes cellular senescence—cells becoming dysfunctional but failing to die, instead producing more inflammatory signals that further damage surrounding tissue.
The pattern is consistent across diseases: chronic inflammation creates cellular and tissue environments favoring disease development, then accelerates disease progression once established. Reducing inflammatory burden doesn’t just make you feel better—it addresses the underlying mechanism driving age-related disease.
What Causes Chronic Inflammation?
Understanding what drives chronic inflammation allows targeted intervention. Multiple factors contribute, often synergistically.
Excess body fat, particularly visceral fat around organs, is perhaps the strongest driver of chronic inflammation. Fat tissue isn’t metabolically inert—it’s an active endocrine organ producing hormones and inflammatory cytokines. Visceral fat specifically is infiltrated with immune cells producing IL-6, TNF-alpha, and other inflammatory molecules.
This explains why obesity so dramatically increases disease risk. The inflammatory state created by excess fat tissue causes insulin resistance, promotes atherosclerosis, and increases cancer risk. Losing visceral fat—measured by waist circumference or DEXA scan—reduces inflammatory markers more than any other single intervention.
Poor diet quality drives inflammation through multiple mechanisms. High intake of refined carbohydrates and added sugars creates glucose and insulin spikes that trigger inflammatory responses. Oxidized vegetable oils high in omega-6 fatty acids (corn, soybean, sunflower oils) promote inflammation, especially when the omega-6 to omega-3 ratio is high. Processed foods containing emulsifiers, preservatives, and other additives may alter gut microbiome and intestinal permeability, triggering immune activation.
Trans fats—still present in some processed foods despite regulations—are particularly inflammatory. Even small amounts measurably increase CRP and other inflammatory markers.
Physical inactivity is independently inflammatory. Exercise has potent anti-inflammatory effects, reducing CRP, IL-6, and TNF-alpha while increasing anti-inflammatory cytokines like IL-10. Sedentary people have chronically elevated inflammatory markers even after controlling for weight. The inflammatory consequences of inactivity contribute to its strong association with chronic disease.
Poor sleep quality and insufficient sleep increase inflammation measurably. Even partial sleep restriction elevates CRP, IL-6, and TNF-alpha within days. Chronic sleep deprivation maintains elevated inflammatory status that never fully resolves. Sleep apnea is particularly inflammatory due to intermittent hypoxia and sleep fragmentation.
Chronic stress activates inflammatory pathways through cortisol dysregulation and sympathetic nervous system activation. Psychological stress measurably increases inflammatory markers. People experiencing chronic stress have elevated CRP and faster cognitive decline, mediated partly through inflammatory mechanisms.
Smoking is profoundly inflammatory, dramatically elevating CRP and other markers. Inflammation is a primary mechanism by which smoking causes cardiovascular disease, cancer, and COPD. Smoking cessation reduces inflammatory markers within months.
Chronic infections can drive ongoing inflammation. Periodontal disease (gum disease) is a significant source of systemic inflammation, with oral bacteria and inflammatory molecules entering circulation. Other chronic infections—H. pylori, chronic viral infections, chronic sinusitis—similarly contribute to inflammatory burden.
Gut dysbiosis and intestinal permeability may contribute to chronic inflammation through mechanisms still being characterized. Disrupted gut microbiome, often from antibiotic use, poor diet, or chronic stress, can increase intestinal permeability (“leaky gut”), allowing bacterial products to enter circulation and trigger immune responses.
Environmental toxins including air pollution, pesticides, heavy metals, and industrial chemicals can trigger inflammatory responses. Air pollution particularly increases cardiovascular disease risk through inflammatory mechanisms.
Aging itself is associated with increasing inflammation—”inflammaging”—as cellular senescence increases, immune regulation becomes impaired, and cumulative damage activates inflammatory pathways. This partly explains why disease risk accelerates with age.
Most people have multiple contributing factors. Addressing chronic inflammation requires identifying your specific drivers and targeting them systematically.
Measuring Inflammation: Key Biomarkers
You can’t manage what you don’t measure. Several biomarkers reveal inflammatory status with varying specificity and clinical utility.
High-sensitivity C-reactive protein (hs-CRP) is the primary clinical marker of systemic inflammation. CRP is produced by the liver in response to inflammatory cytokines, particularly IL-6. Levels correlate with overall inflammatory burden and predict cardiovascular events, diabetes, and mortality.
hs-CRP is measured in mg/L, and interpretation is straightforward:
- Less than 1 mg/L: Low inflammatory risk
- 1-3 mg/L: Moderate inflammatory risk
- Greater than 3 mg/L: High inflammatory risk
Values above 10 mg/L suggest acute inflammation (infection, injury) rather than chronic inflammation and warrant medical evaluation. For assessing chronic inflammatory status, you want hs-CRP measured when you’re healthy, not during acute illness.
hs-CRP is inexpensive, widely available, and well-validated. It should be part of any comprehensive health assessment. Serial measurements over time show whether interventions are reducing inflammation.
Erythrocyte sedimentation rate (ESR) measures how quickly red blood cells settle in a test tube. Faster settling indicates higher levels of inflammatory proteins. ESR less than 20 mm/hr is generally normal, though interpretation varies by age and sex.
ESR is less specific than CRP—many factors affect it—but very elevated values (above 40-50 mm/hr) definitely indicate significant inflammation requiring investigation.
Fibrinogen is a clotting protein that increases during inflammation. Elevated fibrinogen (above 350-400 mg/dL) indicates inflammation and independently predicts cardiovascular events. It’s less commonly measured than CRP but provides additional information about inflammatory status and thrombotic risk.
Interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-alpha) are inflammatory cytokines directly mediating tissue damage. They’re measured in research settings but rarely in clinical practice due to cost and lack of standardized reference ranges. Elevated levels confirm inflammatory state.
Homocysteine is an amino acid that accumulates when B vitamin metabolism is impaired. Elevated homocysteine (above 10-12 μmol/L) increases cardiovascular and cognitive risk partly through inflammatory mechanisms. It also indicates inadequate vitamin B12, B6, or folate status.
Lipoprotein-associated phospholipase A2 (Lp-PLA2) is an inflammatory enzyme specific to vascular inflammation. Elevated Lp-PLA2 (above 200 ng/mL) predicts cardiovascular events independent of cholesterol and CRP. It’s a specialized marker used for cardiovascular risk assessment.
White blood cell count (WBC), while primarily used to detect infection, correlates with chronic inflammation when chronically elevated in the high-normal range (above 7,000-8,000 cells/μL). People with WBC consistently above 8,000 have increased cardiovascular and diabetes risk compared to those below 6,000.
Neutrophil-to-lymphocyte ratio (NLR) is calculated from complete blood count differential. NLR above 2.5-3.0 indicates inflammation and predicts cardiovascular events, cancer mortality, and overall mortality. It’s free—calculated from standard CBC—and surprisingly predictive.
For practical inflammatory status assessment, measure hs-CRP at minimum. Adding homocysteine, fibrinogen, and calculating NLR from routine CBC provides comprehensive inflammatory profiling at reasonable cost. Serial measurement every 6-12 months tracks whether interventions are working.
Evidence-Based Strategies to Reduce Inflammation
Understanding inflammation is useful only if you implement strategies that actually reduce it. Here’s what evidence supports.
Weight Loss and Body Composition Optimization
Losing excess body fat, particularly visceral fat, is the single most effective anti-inflammatory intervention for most people. Every kilogram of fat lost reduces CRP by approximately 0.13 mg/L. Losing 10 kg (22 lbs) can drop CRP by 1.3 mg/L—moving someone from high inflammatory risk to low risk.
The effect is dose-dependent—more weight loss produces greater inflammation reduction. It’s also rapid—CRP decreases within weeks of starting weight loss, before substantial weight change occurs, likely due to improved insulin sensitivity and reduced adipose tissue dysfunction.
Focus particularly on visceral fat reduction through caloric deficit combined with resistance training to preserve muscle mass. DEXA scans tracking visceral fat provide useful feedback beyond just weight.
Anti-Inflammatory Diet Patterns
Diet profoundly affects inflammatory status. Certain dietary patterns consistently reduce inflammation in controlled studies.
A Mediterranean diet emphasizing vegetables, fruits, whole grains, legumes, fish, olive oil, and nuts while limiting red meat and processed foods reduces CRP by 20-30% in intervention studies. The effect is partly from weight loss but remains significant even when calories are controlled.
Omega-3 fatty acids from fatty fish (salmon, mackerel, sardines, herring) or fish oil supplements have direct anti-inflammatory effects, reducing production of inflammatory eicosanoids and cytokines. Aim for 2-3 servings of fatty fish weekly or 2-4g combined EPA/DHA daily from supplements. Studies show CRP reductions of 15-30% with adequate omega-3 intake.
Polyphenol-rich foods including berries, dark leafy greens, green tea, dark chocolate (70%+ cacao), and extra virgin olive oil have anti-inflammatory compounds that reduce oxidative stress and inflammatory signaling. Consistent intake of polyphenol-rich foods correlates with lower inflammatory markers.
Fiber intake particularly from whole grains, legumes, vegetables, and fruits reduces inflammation. High fiber intake (25-30g daily or more) is associated with 20-40% lower CRP compared to low intake. Fiber affects gut microbiome, produces anti-inflammatory short-chain fatty acids, and improves metabolic health.
Foods to minimize or avoid:
- Refined carbohydrates and added sugars elevate glucose and insulin, triggering inflammatory responses
- Vegetable oils high in omega-6 (corn, soybean, sunflower) particularly when omega-3 intake is low
- Processed meats (hot dogs, bacon, deli meats) contain inflammatory preservatives and advanced glycation end products
- Trans fats (partially hydrogenated oils) are profoundly inflammatory even in small amounts
- Fried foods create oxidized fats that promote inflammation
- Excess alcohol (more than 1-2 drinks daily) increases inflammation
The overall pattern matters more than individual foods. A diet dominated by whole foods—vegetables, fruits, legumes, whole grains, fish, nuts, olive oil—with minimal processed foods naturally reduces inflammatory burden.
Regular Exercise
Exercise has potent anti-inflammatory effects independent of weight loss. Both aerobic exercise and resistance training reduce inflammatory markers, though mechanisms differ.
Aerobic exercise reduces CRP by 20-30% with consistent training. Even moderate-intensity exercise (brisk walking 30-45 minutes most days) significantly reduces inflammation. Higher volumes and intensities produce greater effects.
Exercise acutely causes brief inflammatory responses—IL-6 rises during exercise—but this triggers anti-inflammatory responses that dominate afterward, leading to net inflammation reduction. Regular exercisers maintain chronically lower inflammatory markers than sedentary people.
Resistance training reduces inflammation through different pathways, partly by increasing muscle mass. Muscle tissue produces anti-inflammatory myokines that counteract inflammatory cytokines from fat tissue. Preserving and building muscle through resistance training creates an ongoing anti-inflammatory environment.
The recommendation for inflammation reduction: at least 150 minutes weekly of moderate-intensity aerobic exercise or 75 minutes of vigorous exercise, plus resistance training 2-3 times weekly. More is better for inflammation reduction up to a point—excessive training can be pro-inflammatory.
Sleep Optimization
As discussed in previous content, sleep profoundly affects inflammation. Even partial sleep restriction increases CRP, IL-6, and TNF-alpha. Chronic sleep deprivation maintains elevated inflammatory status.
Prioritize 7-9 hours of quality sleep nightly. Address sleep disorders like sleep apnea—treating sleep apnea with CPAP reduces inflammatory markers significantly. Maintain consistent sleep schedules to strengthen circadian rhythm, which regulates inflammatory responses.
Stress Management
Chronic psychological stress elevates inflammatory markers through cortisol dysregulation and autonomic nervous system imbalance. Stress management interventions—meditation, mindfulness, yoga, cognitive behavioral therapy—reduce inflammatory biomarkers in controlled studies.
Even brief daily practices help. Twenty minutes of meditation or deep breathing daily reduces CRP by 15-20% in studies. The key is consistency—occasional stress management doesn’t affect chronic inflammatory status, but daily practice does.
Dental Health
Periodontal disease is a significant source of systemic inflammation. Bacteria from infected gums enter the bloodstream, triggering immune responses and elevating CRP. Treating periodontal disease reduces CRP by 0.5-1.0 mg/L—a clinically significant reduction.
Maintain oral health through twice-daily brushing, daily flossing, and regular dental cleanings. If you have gum disease, get it treated—it’s affecting more than your mouth.
Smoking Cessation
If you smoke, quitting is the single most important intervention you can make for inflammation and overall health. Smoking dramatically elevates CRP and other inflammatory markers. Within months of quitting, inflammatory markers begin declining. Within 1-2 years, inflammation normalizes significantly.
Anti-Inflammatory Supplements
Certain supplements have evidence supporting anti-inflammatory effects, though none replace dietary and lifestyle interventions.
Omega-3 fish oil (2-4g combined EPA/DHA daily) reduces inflammatory markers reliably in studies. Choose quality brands tested for purity.
Curcumin (turmeric extract) has anti-inflammatory properties but poor absorption. Use formulations with piperine (black pepper extract) or phospholipid complexes to enhance absorption. Effective doses are 500-1000mg curcumin daily.
Vitamin D deficiency (below 30 ng/mL) is associated with increased inflammation. Supplementing to achieve levels of 40-60 ng/mL may reduce inflammatory markers in deficient individuals. Typical dosing is 2,000-4,000 IU daily.
Probiotics may reduce inflammation through gut microbiome effects, though evidence is mixed and strain-specific. Multi-strain probiotics (10+ billion CFU daily) show modest anti-inflammatory effects in some studies.
Magnesium deficiency increases inflammation. Adequate intake (400-500mg daily) may reduce CRP in deficient individuals.
Supplements should complement, not replace, dietary and lifestyle anti-inflammatory strategies.
Monitoring Progress: Tracking Inflammatory Status
Implementing anti-inflammatory strategies is only useful if they actually reduce your inflammatory burden. Serial biomarker testing tracks progress.
Get baseline inflammatory markers—at minimum hs-CRP, ideally also homocysteine, fibrinogen, and CBC for NLR calculation. Implement dietary, exercise, sleep, and stress management interventions consistently for 3-6 months, then retest.
You should see measurable improvement. CRP should decrease by at least 20-30% if you’re implementing multiple interventions effectively. If it doesn’t improve, either compliance isn’t as good as you think, or you haven’t addressed your primary inflammatory drivers.
Common scenarios:
CRP remains elevated despite weight loss and exercise—consider gut health issues, chronic infections (particularly dental), or sleep disorders. Address these systematically.
CRP improves but remains above 1.0 mg/L—continue current interventions and add others. Increase omega-3 intake, optimize sleep further, add stress management if not already implemented.
CRP normalizes (below 1.0 mg/L)—maintain current interventions. Retest annually to ensure inflammatory status remains low. Consider retesting every 6 months if making ongoing changes or addressing specific health issues.
Track trends over time rather than obsessing over single values. Occasional transient elevations happen with acute stress, minor infections, or intense exercise. The pattern over months to years matters most.
When to Seek Medical Evaluation
Significantly elevated inflammatory markers warrant medical evaluation to rule out underlying conditions requiring treatment.
hs-CRP persistently above 10 mg/L suggests acute inflammation—infection, autoimmune disease, or other inflammatory conditions requiring diagnosis and treatment. Don’t assume lifestyle causes at this level.
hs-CRP between 3-10 mg/L that doesn’t respond to lifestyle interventions warrants investigation. This could indicate chronic infections, autoimmune conditions, or other inflammatory diseases.
Rapidly rising inflammatory markers over months despite stable lifestyle merit evaluation.
Certain symptoms combined with elevated inflammation require prompt assessment: unexplained weight loss, persistent fever, night sweats, joint pain and stiffness, chronic fatigue not explained by sleep or lifestyle factors.
Most people with chronically elevated CRP in the 1-5 mg/L range have lifestyle-driven inflammation that responds to diet, exercise, weight loss, and sleep optimization. But outliers need medical evaluation to ensure nothing is being missed.
The Bottom Line on Inflammation
Chronic inflammation isn’t a vague concept or wellness trend. It’s the measurable, quantifiable common pathway driving diabetes, cardiovascular disease, cancer, neurodegeneration, and accelerated aging.
You can measure your inflammatory status through simple, inexpensive blood tests. You can track whether it’s improving or worsening over time. And most importantly, you can reduce inflammatory burden through evidence-based lifestyle interventions—weight loss, anti-inflammatory diet, regular exercise, adequate sleep, and stress management.
This isn’t alternative medicine or unproven theory. It’s fundamental physiology supported by overwhelming evidence. Reducing chronic inflammation doesn’t just reduce abstract disease risk—it addresses the mechanism actively driving disease development right now.
For anyone serious about disease prevention and longevity, managing inflammation is non-negotiable. Test your inflammatory biomarkers. Implement interventions targeting your specific drivers. Retest to confirm improvement. Maintain an anti-inflammatory lifestyle long-term.
The compounding benefits over decades are profound. Lower inflammation means reduced diabetes risk, less cardiovascular disease, slower cognitive decline, better immune function, and longer healthspan. This is optimization at the fundamental level where it matters most.

Comprehensive Inflammatory Assessment at Preamble Health
At Preamble Health in Scottsdale, inflammatory status is a key component of our comprehensive health assessments. Our Medicine 3.0 Executive Physical includes extensive inflammatory biomarker testing—hs-CRP, homocysteine, fibrinogen, and comprehensive metabolic panels revealing metabolic inflammation.
We identify your specific inflammatory drivers through detailed assessment of body composition (visceral fat via DEXA), metabolic health (insulin resistance, glucose dysregulation), cardiovascular risk factors, sleep quality, and lifestyle patterns.
Our Core Membership provides ongoing support implementing anti-inflammatory lifestyle interventions with serial biomarker testing tracking progress. We adjust strategies based on your results, ensuring interventions actually reduce inflammatory burden.
This is preventative medicine targeting the root cause of chronic disease, not just managing symptoms after disease develops.
- Schedule comprehensive inflammatory assessment including biomarker testing
- Learn about our Medicine 3.0 Executive Physical with complete metabolic and inflammatory profiling
- Explore our Core Membership for ongoing anti-inflammatory lifestyle support
- Book a consultation to discuss your inflammatory status and optimization strategies